| | 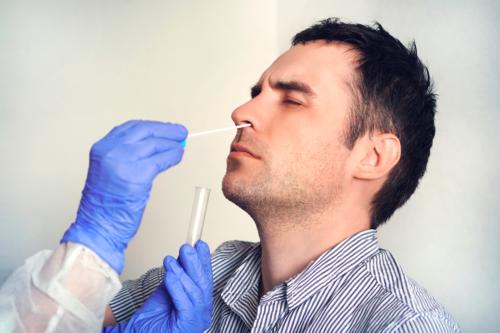
Same Day Appointments Available - Results in 15 mins.
The COVID-19 Antigen (Ag) Rapid Test Device is an in vitro diagnostic rapid test device for the qualitative detection of SARS-CoV-2 antigen (Ag) in human nasopharyngeal swab specimens from individuals who meet COVID-19 clinical and or epidemiological criteria. This test is used as an aid in the diagnosis of SARS-CoV-2 infection.
|
| | |  We offer both Abbots & Roche ECLIA test We offer both Abbots & Roche ECLIA test
This test is a venous blood draw test, which gets sent off for analysis.
IgG antibody testing can tell whether a person has been previously infected, as the host immune system reacts to the infection by SARS-CoV-2 by producing antibodies from a few days to two weeks after the onset of symptoms. Specific IgG antibodies are produced in the later stages of infection to SARS-CoV-2, and are detectable after RNA is no longer detectable.
The persistence of IgG antibodies allows identification of people who have been infected by SARS-CoV-2.
This test has a minimum sensitivity of 98% and a specificity of 100%, with no cross-reactivity with other viruses.
IgM Antibody Testing can give indication of current infection.
|
|
| £ 129.00 (-23.26%) £ 99.00 | 
This test is a Loop mediated isothermal amplification (LAMP) using : Nicking Enzyme Amplification Reaction (NEAR) is a method for in vitro DNA / RNA amplification like the polymerase chain reaction (PCR). NEAR is isothermal, replicating DNA/RNA at a constant temperature using a polymerase (and nicking enzyme) to exponentially amplify the DNA/RNA at a temperature range of 55 °C to 59 °C.
This COVID-19 assay is a rapid molecular in vitro diagnostic test utilizing an isothermal nucleic acid amplification technology intended for the qualitative detection of nucleic acid from the SARS-CoV-2 viral RNA in direct nasal, nasopharyngeal or throat swabs from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms.
|
| | £ 129.00 (-23.26%) £ 99.00 | 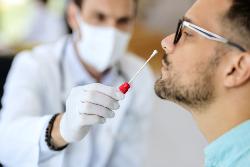
"Fit to Fly " and General Testing.
Results usually in 2-4 hours - Same Day Appointments Available.
PCR (polymerase chain reaction) samples can come from several different sites in the patient. The simplest is the nasal swab taken from well inside the nose. The back of the throat is another option.
To find evidence of the virus, researchers use PCR to copy and amplify any segments of viral genetic code found in the sample, which makes it easier to detect. This typically involves adding reagents and enzymes, and raising and lowering the temperature of the mixture, known as thermal cycling.
Antigen testing reveals whether someone has a current infection and could therefore pass Covid-19 on to others. PCR is the gold standard in viral diagnostics.
|
|
| | 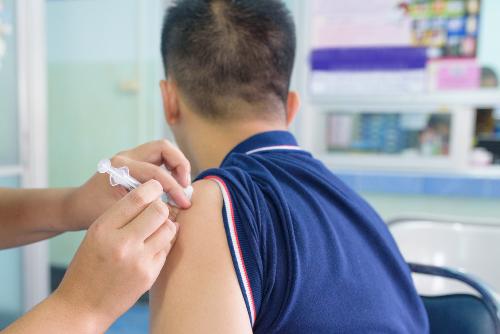
The Anti-SARS-CoV-2-S test - It tests for Antibodies against spike proteins and is generally requested post vaccination. The SCOV provides both a Qualitative result(i.e. Positive or Negative) and a Quantitative result (anything >= 0.8 is positive, anything <0.8 is negative.) This is a serum based test. |
| | |  This test identify the presence of influenza A and B viral nucleoprotein antigens in respiratory specimens, and display the result in a qualitative way (positive vs. negative). This test identify the presence of influenza A and B viral nucleoprotein antigens in respiratory specimens, and display the result in a qualitative way (positive vs. negative). |
|
| |